Unusual thermal phase transition behavior of an ionic liquid and poly(ionic liquid) in water with significantly different LCST and dynamic mechanism†
Abstract
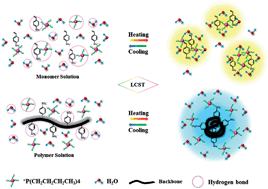
Dynamic thermal phase transition behavior of a well-defined thermoresponsive ionic liquid (IL), [P4,4,4,4][SS], and its polymer, poly(ionic liquid) (PIL), P[P4,4,4,4][SS], synthesized by modified free radical polymerization was investigated by means of DSC, turbidity, temperature-variable 1H NMR, FT-IR spectroscopy measurements, in combination with perturbation correlation moving window (PCMW) and two-dimensional correlation (2Dcos) analysis. Obvious distinction was observed between the LCST of monomer and polymer solutions indicating their largely different dynamic transition process. Further studies revealed that the aggregation of [P4,4,4,4][SS] is mainly driven by the synergetic variations of cations and anions while the separation process of the P[P4,4,4,4][SS] solution is found to be dominated by conformation changes of anions. Finally, 2Dcos was employed to elucidate the sequence order of chemical group motion during heating. It is concluded that synergetic variations of cations and anions in the [P4,4,4,4][SS] solution resulted in the randomly aggregated structure of globules during separation. In contrast, in the P[P4,4,4,4][SS] solution, anions of repeated units tend to be wrapped into the aggregated globules while cations would distribute at the periphery of globules after transition. Additionally, several unusual phenomena were also interpreted, such as the different varying tendency in LCST of monomer and polymer solutions with increasing concentration, their distinction in the final transmittance and their rather different LCST at the same concentration.

 Please wait while we load your content...
Please wait while we load your content...