Trehalose-functionalized block copolymers form serum-stable micelles†
Abstract
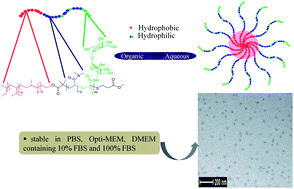
Well-defined amphiphilic diblock terpolymers of poly(ethylene-alt-propylene)–poly[(N,N′-dimethylacrylamide)-grad-poly(6-deoxy-6-methacrylamido trehalose)] (denoted as PEP–poly(DMA-grad-MAT, or PT) have been synthesized using a PEP macromolecular chain transfer agent by reversible addition–fragmentation chain transfer (RAFT) polymerization. The content of MAT was varied from 5 to 14 mole percent in the hydrophilic block of the amphiphilic polymers yielding a family of diblock terpolymers. The reactivity ratios of DMA and trimethyl silyl protected MAT were determined to be r1 = 0.09 ± 0.01 and r2 = 1.6 ± 0.1 and thus, the hydrophilic block was a gradient copolymer consisting of a higher DMA ratio (closer to the PEP) that was terminated with a higher MAT ratio. The silyl groups were subsequently deprotected and micellar dispersions were prepared by two different techniques: nanoprecipitation (NP) and direct dissolution (DD). Micelles formed by the two methods were characterized by dynamic light scattering (DLS) and cryo-transmission electron microscopy (cryo-TEM). These PT diblock terpolymers self-assembled into spherical nanostructures in aqueous media with hydrodynamic radii of ca. 16 nm in the dispersions formed by nanoprecipitation. Micellar dispersions with greater MAT content (11 mole percent in the hydrophilic block) exhibited sufficient contrast in cryoTEM images and the corona were clearly seen as gray halos around the micellar cores. The stability of each micellar dispersion in different biological media was examined over a period of 14 h using DLS. These results indicated that micellar dispersions are stable in 100 percent fetal bovine serum (FBS), which offer promise for in vivo actives delivery applications.

 Please wait while we load your content...
Please wait while we load your content...