In vitro and in vivo application of hydroxypropyl-β-cyclodextrin-grafted polyethyleneimine used as a transdermal penetration enhancer
Abstract
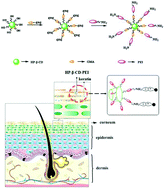
Biodegradable cationic polymers have a promising application in transdermal drug delivery. In this study, hydroxypropyl-β-cyclodextrin-grafted polyethyleneimine (HP-β-CD–PEI) were prepared, and these polymers were developed as a penetration enhancer. The aim of this study was to evaluate the in vitro and in vivo application of the transdermal permeation enhancement of HP-β-CD–PEI with different molecular weights (Mol. Wt) of polyethyleneimine (PEI). HP-β-CD–PEI with different Mol. Wt of PEI (600, 1.8 kDa and 10 kDa) were synthesized and characterized by 1H NMR. The effect of HP-β-CD–PEI on the stratum corneum was studied by attenuated total reflection-Fourier transform infrared spectroscopy (ATR-FTIR). In vitro prostaglandin E2 (PGE2) release by cell culture, and skin morphological changes were determined to examine the irritation of the skin by HP-β-CD–PEI. Vitamin B12 was chosen as a model drug to examine the enhancing capacity of this enhancer. Both in vitro permeation by Franz cells and in vivo kinetics of skin disposition were performed to determine the drug permeation by HP-β-CD–PEI. The drug with the enhancer was penetrated through mice skin for 3, 6, and 9 h, and the cumulative drug content deposited in the epidermis and dermis was measured. The results showed that the HP-β-CD–PEI could be a promising enhancer for topical drug delivery.

 Please wait while we load your content...
Please wait while we load your content...