Topochemical polymerization using bis-thyminyl monomers†
Abstract
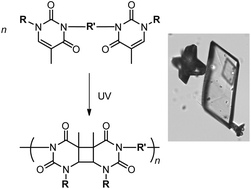
Topochemical polymerization is one way to synthesize novel macromolecular architectures with stereoregular chain structures. The [2π + 2π]-cycloaddition of certain conjugated diolefin monomers is one such form of topochemical polymerization. Unfortunately, the design of new monomers that can undergo topochemical [2π + 2π]-polymerization to form linear polymers can be a formidable challenge due to the strict geometrical criteria governing the reactivity of monomer crystals. In this research ten bis-thymine monomers were synthesized, that varied with respect to the N1-functionality, the size of the N3–N3-polymethylene spacer, or the type of rigid N3–N3-aryl spacer employed. On irradiation, five of the crystalline monomers were found to undergo topochemical photo-reactions. The crystal structures of the bis-thyminyl monomers were used to investigate the monomer structure–reactivity relationships. The molecular weights of the photo-products were determined and the thermal properties of some of the new materials were also evaluated using thermo analytical techniques.

 Please wait while we load your content...
Please wait while we load your content...