Fully biodegradable antibacterial hydrogels via thiol–ene “click” chemistry†
Abstract
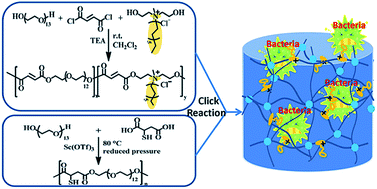
In this work, fully biodegradable antimicrobial hydrogels were prepared facilely via a thiol–ene “click” reaction under human physiological conditions using multifunctional poly(ethylene glycol) (PEG) derivatives as precursors. Water soluble and degradable PEG derivatives with multi-enes and multi-thiols, respectively, were synthesized by polycondensation of oligo(ethylene glycol) (OEG) with “clickable” monomers. Ammonium groups with long alkyl chains were incorporated into one of the precursors covalently, using dodecyl bis(2-hydroxyethyl) methylammonium chloride as a comonomer. Proton nuclear magnetic resonance (1H-NMR) spectroscopy, gel permeation chromatography (GPC) and Fourier transform infrared spectroscopy (FT-IR) were used to characterize the precursors and hydrogels. These types of cationic PEG-type hydrogels showed strong antibacterial abilities against both Gram-negative and Gram-positive bacteria due to the ammonium moieties. Moreover, the hydrogel with fewer ammonium moieties still possessed significant antibacterial abilities, but low toxicity, and has the potential to be used as a medical material.

 Please wait while we load your content...
Please wait while we load your content...