Elaboration of drug nanocarriers based on a glucosamine labeled amphiphilic polymer†
Abstract
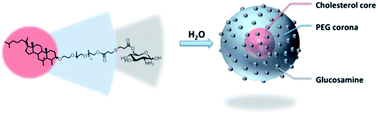
A new functional polymer micelle with high loading efficiency of a poorly soluble drug was made of biocompatible and/or biosourced compounds, i.e. cholesterol–poly(ethylene glycol)–glucosamine (Chol–PEG–GlcNH2). A synthesis strategy combining enzymatic and metal-free click chemistry was developed in order to meet the increasingly stringent requirements of biomedical applications. After the self-assembly of the Chol–PEG–GlcNH2 amphiphilic polymer in water, the presence of glucosamine at the micelle surface confers an active targeting moiety to the nanocarriers whereas protonation of the peripheral primary amine delay their aggregation. The complete characterization of this novel functional amphiphilic bioconjugate is presented as well as its aqueous solution behaviour and encapsulation efficiency using ketoconazole as a model hydrophobic drug.

 Please wait while we load your content...
Please wait while we load your content...