Synthesis of fluorinated analogues of a practical polymer TQ for improved open-circuit voltages in polymer solar cells†
Abstract
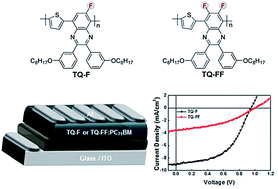
In an attempt to further lower the HOMO of a cost-effective polymer poly(2,3-bis-(3-octyloxyphenyl)quinoxaline-5,8-dyl-alt-thiophene-2,5-diyl) (TQ) by adding F atoms onto the existing quinoxaline acceptor within the polymer backbone, we have synthesized two structurally identical fluorinated analogues of TQ (TQ-F (single F) and TQ-FF (double F)), except for the number of F atoms. The effects of inclusion of F atoms on the optical properties, nature of charge transport, and molecular organization are thoroughly investigated. The resulting two fluorinated polymers show a decrease in both the HOMO and the LUMO energy levels relative to non-fluorinated TQ. Moreover, the fluorination of the polymer backbone has lowered the HOMOs more than the LUMOs, slightly widening the energy bandgaps as the number of F atoms increases. Thus, use of these polymers in bulk-heterojunction (BHJ) solar cells, in all cases, leads to large VOC values. The power conversion efficiency (PCE) of the optimized PSCs based on TQ-F reaches 4.41%. In addition, it is interesting to note that, despite TQ-FF having the PCE that is lower than that of TQ-F, an unprecedentedly high VOC of 1.00 V is achieved, which is nearly equal to the highest VOC values ever reported for polymers.

 Please wait while we load your content...
Please wait while we load your content...