Synthesis and characterization of thermo-responsive polypeptoid bottlebrushes†
Abstract
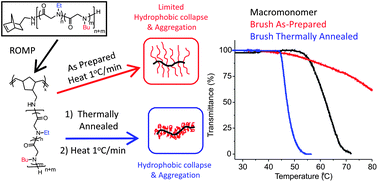
Linear polypeptoid macromonomers with controlled chain length and composition were prepared by norbornene methylamine-initiated copolymerization of N-ethyl N-carboxyanhydride (Et-NCA) and N-butyl N-carboxyanhydride (Bu-NCA). Polypeptoid bottlebrush copolymers with varying backbone length and side chain composition were subsequently synthesized using the grafting-through method via the ring-opening metathesis polymerization (ROMP) of norbornenyl-terminated poly(N-ethyl glycine)-ran-poly(N-butyl glycine) macromonomers. The bottlebrush copolypeptoids are thermally responsive with a cloud point transition in aqueous solution similar to the linear macromonomers. In contrast to the linear macromonomers, the cloud point transition of the bottlebrush copolypeptoids is strongly dependent on the thermal history of the solution. Freshly prepared aqueous solutions of the bottlebrush copolypeptoids do not exhibit any notable cloud point transition, whereas the solutions that have been thermally annealed at high temperature exhibit sharp and reversible cloud point transition. Intriguingly, upon inorganic salt addition, the aqueous solutions of the bottlebrush copolypeptoids exhibit normal cloud point transition which is independent of the solution thermal history. It was suggested that the thermal annealing and the presence of a salt facilitated the conformational reorganization of the bottlebrush copolypeptoids to favor hydrophobic collapse and intermolecular aggregation, resulting in a cloud point transition.

 Please wait while we load your content...
Please wait while we load your content...