Bringing d-limonene to the scene of bio-based thermoset coatings via free-radical thiol–ene chemistry: macromonomer synthesis, UV-curing and thermo-mechanical characterization†
Abstract
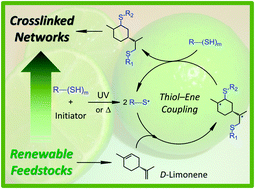
The increasing pursuit for bio-based plastic materials led us to investigate the potential use of the monoterpene limonene in thermoset synthesis using the free-radical mediated thiol–ene reaction. The high efficiency of this reaction to prepare multifunctional ene-terminated resins, as intermediary macromolecular precursors, for thermosets synthesis was demonstrated under thermal and photoinitiated conditions. Although an excess of terpene favors formation of well-defined macromonomers in organic solution, the characteristic low-vapor pressure of limonene hinders its simple removal (or recycling) via evaporation after synthesis. Alteration to an initial thiol–ene stoichiometry of 1 : 0.5 enables production of high molecular weight resins in the form of ‘hyperbranched oligomeric-like’ structures having moderate polydispersity. UV-curing of these polyfunctional resins combined with equal mole compositions of multifunctional alkyl ester 3-mercapto propionates yields highly sticky, amorphous and flexible elastomers with different thermo-mechanical properties. These can be further modulated by varying the amount of unreacted thiol occluded within the networks working as a plasticizer. Introduction of a renewable cycloaliphatic structure into the materials offers a convenient way to enhance the glass-transition temperature and stiffness of traditional thiol–ene networks. The materials synthesized may be considered potentially useful as sealants and adhesives in a wide variety of applications including organic coatings. The versatility of UV-irradiation over thermal initiation makes this method particularly suitable for green industrial synthesis processes via thiol–ene chemistry using limonene and multifunctional thiols. The thiol–ene system evaluated herein serves as a model example for the sustainable incorporation of natural diolefinic monomers into semi-synthetic thiol–ene networks exhibiting a range of thermo-mechanical properties.
- This article is part of the themed collection: Sustainable polymers: replacing polymers derived from fossil fuels

 Please wait while we load your content...
Please wait while we load your content...