One-pot synthesis and gelation by borax of glycopolymers in water†
Abstract
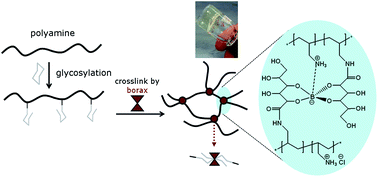
A glycosylated polyamine is synthesized using a straightforward route in aqueous solution. An optimum of grafting is obtained when equimolar amounts of gluconolactone and NaOH are added to an aqueous solution of poly(allylamine hydrochloride), PAH. Under these conditions, up to 60% of gluconolactone is grafted onto the PAH chain, the remaining being transformed into gluconate. The very fast grafting reaction, completed in less than 5 minutes at room temperature, may be applicable to a large range of water-soluble polymers bearing amino groups. Upon addition of borax before any purification process, the reaction mixture can be transformed in situ into a hydrogel. The gelation is attributed to the formation of borate diesters acting as cross-linkers of a number of PAH chains bearing glycosylated grafts. The viscoelastic properties of the gels can be triggered by the amounts of gluconolactone and borax incorporated in the mixture as well as by the final pH. Owing to the remaining ammonium groups along the chains which stabilize the borate complex, gels can be obtained at physiological pH, a performance superior to those obtained with comparable poly(vinyl alcohol)- or guar/borax systems.

 Please wait while we load your content...
Please wait while we load your content...