Thermoplastic polyesters and Co-polyesters derived from vegetable oil: synthesis and optimization of melt polycondensation for medium and long chain poly(ω-hydroxyfatty acid)s and their ester derivatives†
Abstract
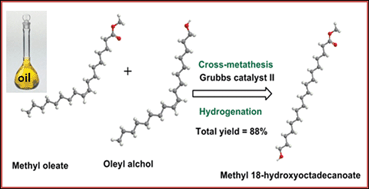
The synthesis of a series of ω-hydroxyfatty acid (ω-OHFA) monomers and their methyl ester derivatives (Me-ω-OHFA) from mono-unsaturated fatty acids and alcohols via ozonolysis-reduction/cross-metathesis reactions is described. Melt polycondensation of the monomers yielded thermoplastic poly(ω-hydroxyfatty acid)s [–(CH2)n–COO–]x with medium (n = 8 and 12) and long (n = 17) repeating monomer units. The ω-OHFAs and Me-ω-OHFAs were all obtained in good yield (≥80%) and purity (≥97%) as established by 1H NMR, Fourier Transform infra-red spectroscopy (FT-IR), mass spectroscopy (ESI-MS) and high performance liquid chromatography (HPLC) analyses. The average molecular size (Mn) and distribution (PDI) of the poly(ω-hydroxyfatty acid)s (P(ω-OHFA)s) and poly(ω-hydroxyfatty ester)s (P(Me-ω-OHFA)s) as determined by GPC varied with organo-metallic Ti(IV) isopropoxide [Ti(OiPr)4] polycondensation catalyst amount, reaction time and temperature. An optimization of the polymerization process provided P(ω-OHFA)s and P(Me-ω-OHFA)s with Mn and PDI values desirable for high end applications. Co-polymerization of the long chain (n = 12) and medium chain (n = 8) Me-ω-OHFAs by melt polycondensation yielded poly(ω-hydroxy tridecanoate/ω-hydroxy nonanoate) random co-polyesters (Mn = 11 000–18 500 g mol−1) with varying molar compositions.
- This article is part of the themed collection: Sustainable polymers: replacing polymers derived from fossil fuels

 Please wait while we load your content...
Please wait while we load your content...