Subcomponent self-assembly of polymer chains based on dynamic and geometrical coordination diversity of the first row transition metal ions†
Abstract
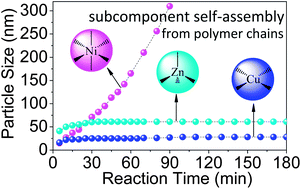
The subcomponent self-assembly of polymer chains based on the dynamic/geometrical coordination diversity of first row transition metal ions is reported. A well-defined NH2-functional hydrophilic block copolymer, Ni(II), Cu(II) and Zn(II) and a salicylaldehyde derivative are selected as subcomponents. The imine formation and self-assembly are followed using 1H NMR, DLS, GPC, and UV-vis spectroscopy. The results demonstrate that the subcomponent self-assembly strongly depends on the coordination stability constants and geometries of these metal ions. Ni(II) induces interchain coordination, and rapid phase transition to macro-phase separation but with the lowest autocatalysis efficiency. However, Cu(II) ions induce intrachain coordination with the most remarkable autocatalysis, and the delaying of the phase transition to form well-defined hybrid nanoparticles. The behaviour with Zn(II) ions is in between that with Ni(II) and Cu(II) ions. This striking differentiation demonstrates that the dynamic/geometrical coordination diversity of the abundant first row transition metal ions effectively control the subcomponent self-assembly of polymer chains, and thus the hierarchical nanostructures of dative metal–polymer hybrid functional materials.

 Please wait while we load your content...
Please wait while we load your content...