Post-metalation of porous aromatic frameworks for highly efficient carbon capture from CO2 + N2 and CH4 + N2 mixtures†
Abstract
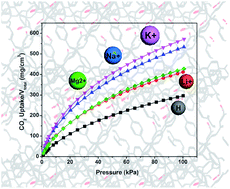
The development of microporous materials for carbon capture, especially for carbon dioxide and methane, is a rapidly growing field based on the increasing demand for clean energy and pressing environmental concerns of global warming effected by greenhouse gases. To achieve this goal of developing carbon selective porous materials, a new porous aromatic framework featuring carboxyl-decorated pores, PAF-26-COOH, has been synthesized successfully. The modification of PAF-26 materials with representative light metals is exemplified by Li, Na, K and Mg via a post-metalation approach. The obtained PAF-26 products exhibit moderate surface area and controllable pore size at the atomic level. Gas sorption of CO2, CH4 and N2 is carried out on as-prepared PAF-26 samples at mild temperatures (273 K and 298 K). It is found that the PAF-26 materials show high adsorption capacity for CO2 and CH4 and low ability toward N2. Particularly, as-synthesized PAF-26 compounds exhibit remarkably high isosteric heats of adsorption toward CO2 and CH4, indicating high affinity for CO2 and CH4 gases. The gas selectivity for CO2–N2 and CH4–N2 mixtures is predicted by the IAST model. High selectivity of 80 for CO2 over N2 is obtained for PAF-26-COOMg. In addition, high selectivity values of CH4 over N2 are observed. The high performance including high storage capacity and selectivity makes PAF-26 materials promising for carbon capture or sequestration.

 Please wait while we load your content...
Please wait while we load your content...