Iridium-catalyzed C–H borylation of pyridines†
Abstract
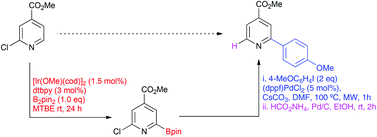
The iridium-catalysed C–H borylation is a valuable and attractive method for the preparation of aryl and heteroaryl boronates. However, application of this methodology for the preparation of pyridyl and related azinyl boronates can be challenged by low reactivity and propensity for rapid protodeborylation, particularly for a boronate ester ortho to the azinyl nitrogen. Competition experiments have revealed that the low reactivity is due to inhibition of the active catalyst through coordination of the azinyl nitrogen lone pair at the vacant site on the iridium. This effect can be overcome through the incorporation of a substituent at C-2. Moreover, when this is sufficiently electron-withdrawing protodeborylation is sufficiently slowed to permit isolation and purification of the C-6 boronate ester. Following functionalization, reduction of the directing C-2 substituent provides the product arising from formal ortho borylation of an unhindered pyridine ring.

 Please wait while we load your content...
Please wait while we load your content...