Recent developments in transition metal-catalysed spiroketalisation
Abstract
The spiroketal motif occurs in a wide range of biologically active natural products and represents a valuable target in medicinal chemistry and total synthesis. In recent years, innovative new synthetic methods have substantially expanded the range of potential precursors, cyclisation modes and opportunities for asymmetric catalysis and tandem processes. This Perspective aims to highlight recent rapid advances in the use of transition metal catalysis for spiroketal formation, in the context of our own investigations into gold-catalysed asymmetric spiroketalisation.
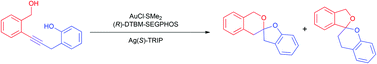

 Please wait while we load your content...
Please wait while we load your content...