Monitoring penetratin interactions with lipid membranes and cell internalization using a new hydration-sensitive fluorescent probe†
Abstract
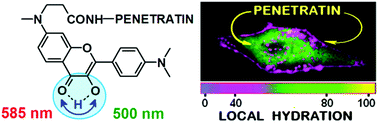
A new fluorescent label N-[4′-(dimethylamino)-3-hydroxyflavone-7-yl]-N-methyl-β-alanine (7AF) was synthesized. Due to two electron donor groups at the opposite ends of the chromophore, an excited state intramolecular proton transfer (ESIPT) resulting in a dual emission was observed even in highly polar media and its fluorescence quantum yield was found to be remarkably high in a broad range of solvents including water. As a consequence, this label exhibits a remarkable sensitivity to the hydration of its environment, which is observed as a color switch between the emission of the ESIPT product (T* form) and that of the normal N* form. The 7AF label was coupled to the N-terminus of penetratin, a cell penetrating peptide, in order to study its interactions with lipid membranes and internalization inside the cells. As expected, the binding of penetratin to lipid membranes resulted in a dramatic switch in the relative intensity of its two emission bands as compared to its emission in buffer. Our studies with different lipid compositions confirmed the preference of penetratin to lipid membranes of the liquid disordered phase. After incubation of low concentrations of labeled penetratin with living cells, ratiometric imaging revealed, in addition to membrane-bound species, a significant fraction of free peptide in cytosol showing the characteristic emission from aqueous medium. At higher concentrations of penetratin, mainly peptides bound to cell membrane structures were observed. These observations confirmed the ability of penetratin to enter the cytosol by direct translocation through the cell plasma membrane, in addition to the classical entry by endocytosis. The present probe constitutes thus a powerful tool to study the interaction of peptides with living cells and their internalization mechanisms.

 Please wait while we load your content...
Please wait while we load your content...