Organic synthetic transformations using organic dyes as photoredox catalysts
Abstract
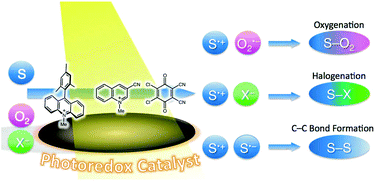
The oxidizing ability of organic dyes is enhanced significantly by photoexcitation. Radical cations of weak electron donors can be produced by electron transfer from the donors to the excited states of organic dyes. The radical cations thus produced undergo bond formation reactions with various nucleophiles. For example, the direct oxygenation of benzene to phenol was made possible under visible-light irradiation of 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ) in an oxygen-saturated acetonitrile solution of benzene and water via electron transfer from benzene to the triplet excited state of DDQ. 3-Cyano-1-methylquinolinium ion (QuCN+) can also act as an efficient photocatalyst for the selective oxygenation of benzene to phenol using oxygen and water under homogeneous and ambient conditions. Alkoxybenzenes were also obtained when water was replaced by alcohol under otherwise identical experimental conditions. QuCN+ can also be an effective photocatalyst for the fluorination of benzene with O2 and fluoride anion. Photocatalytic selective oxygenation of aromatic compounds was achieved using an electron donor–acceptor-linked dyad, 9-mesityl-10-methylacridinium ion (Acr+–Mes), as a photocatalyst and O2 as the oxidant under visible-light irradiation. The electron-transfer state of Acr+–Mes produced upon photoexcitation can oxidize and reduce substrates and dioxygen, respectively, leading to the selective oxygenation and halogenation of substrates. Acr+–Mes has been utilized as an efficient organic photoredox catalyst for many other synthetic transformations.

 Please wait while we load your content...
Please wait while we load your content...