Complexity generation by chemical synthesis: a five-step synthesis of (−)-chaetominine from l-tryptophan and its biosynthetic implications†‡
Abstract
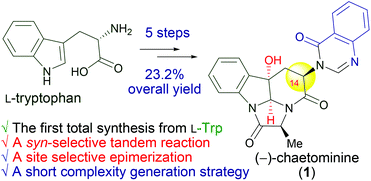
We demonstrated, for the first time, that on the basis of chemistry principles, the hexacyclic peptidyl alkaloid (−)-chaetominine (1) can be synthesized in a straightforward manner from L-Trp. The approach features the efficient generation of molecular complexity via a tandem C3/C14 syn-selective epoxidation (dr = 3 : 2)–annulative ring-opening reaction and a regioselective epimerization at C14. The successful production of (−)-chaetominine (1) from L-Trp could be helpful for revealing how the configuration of L-tryptophan becomes inverted in the biosynthetic pathway of (−)-chaetominine (1).

 Please wait while we load your content...
Please wait while we load your content...