Tuning nondoped carbon nanotubes to an efficient metal-free electrocatalyst for oxygen reduction reaction by localizing the orbital of the nanotubes with topological defects†
Abstract
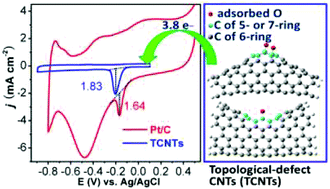
Breaking the electron delocalization of sp2 carbon materials by heteroatom doping is a practical strategy to produce metal-free electrocatalysts of oxygen reduction reaction (ORR) for fuel cells. Whether carbon nanotubes (CNTs) can be efficiently tuned into ORR electrocatalysts only by intrinsic defects rather than heteroatom doping has not been well studied yet in experiment and theory. Here we introduce topological defects of nonhexagon carbon rings into CNTs to break the delocalization of their orbitals and make such type of CNTs to be a high-performance ORR catalyst. The electrochemical tests and theoretical studies indicate that the O2 chemisorption and the following electrocatalytic activity are promoted by the introduced topological defects and show a strong dependence on the defect amount. Such topological-defect CNTs (TCNTs) have an excellent ORR performance owing to a 3.8-electron-transferring process, ∼4 times higher current density and ∼120 mV more positive peak potential than normally straight CNTs. Moreover, TCNTs show a higher steady-state diffusion current density and much better stability and immunity to crossover effect as compared with commercial Pt/C catalyst. Hence, our results strongly suggest that tuning the surface structure of CNTs with nonhexagon carbon rings is a novel strategy for designing advanced ORR electrocatalysts for fuel cells.

 Please wait while we load your content...
Please wait while we load your content...