A facile and versatile method for preparation of colored TiO2 with enhanced solar-driven photocatalytic activity†
Abstract
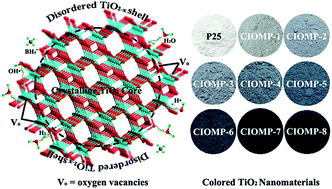
Colored TiO2 has attracted enormous attention due to its visible light absorption and excellent photocatalytic activity. In this report, we develop a simple and facile solid-state chemical reduction approach for a large-scale production of colored TiO2 at mild temperature (300–350 °C). The obtained sample possesses a crystalline core/amorphous shell structure (TiO2@TiO2−x). The oxygen vacancy results in the formation of a disordered TiO2−x shell on the surface of TiO2 nanocrystals. XPS and theoretical calculation results indicate that valence band tail and vacancy band below the conduction band minimum appear for the TiO2−x, which implies that the TiO2@TiO2−x nanocrystal has a narrow band gap and therefore leads to a broad visible light absorption. Oxygen vacancy in a proper concentration promotes the charge separation of photogenerated carriers, which improves the photocatalytic activity of TiO2@TiO2−x nanocrystals. This facile and general method could be potentially used for large scale production of colored TiO2 with remarkable enhancement in the visible light absorption and solar-driven H2 production.

 Please wait while we load your content...
Please wait while we load your content...