Large work function difference driven electron transfer from electrides to single-walled carbon nanotubes†
Abstract
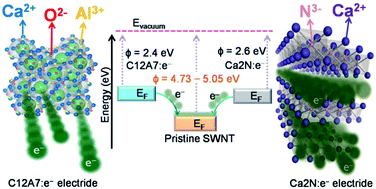
A difference in work function plays a key role in charge transfer between two materials. Inorganic electrides provide a unique opportunity for electron transfer since interstitial anionic electrons result in a very low work function of 2.4–2.6 eV. Here we investigated charge transfer between two different types of electrides, [Ca2N]+·e− and [Ca24Al28O64]4+·4e−, and single-walled carbon nanotubes (SWNTs) with a work function of 4.73–5.05 eV. [Ca2N]+·e− with open 2-dimensional electron layers was more effective in donating electrons to SWNTs than closed cage structured [Ca24Al28O64]4+·4e− due to the higher electron concentration (1.3 × 1022 cm−3) and mobility (∼200 cm2 V−1 s−1 at RT). A non-covalent conjugation enhanced near-infrared fluorescence of SWNTs as high as 52%. The field emission current density of electride–SWNT–silver paste dramatically increased by a factor of 46 000 (14.8 mA cm−2) at 2 V μm−1 (3.5 wt% [Ca2N]+·e−) with a turn-on voltage of 0.85 V μm−1.

 Please wait while we load your content...
Please wait while we load your content...