Can small hydrophobic gold nanoparticles inhibit β2-microglobulin fibrillation?†
Abstract
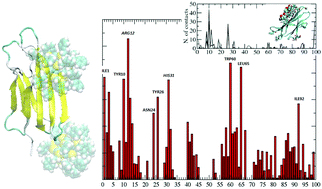
Inorganic nanoparticles stabilized by a shell of organic ligands can enhance or suppress the natural propensity of proteins to form fibrils. Functionalization facilitates targeted delivery of the nanoparticles to various cell types, bioimaging, drug delivery and other therapeutic and diagnostic applications. In this study, we provide a computational model of the effect of a prototypical thiol-protected gold nanoparticle, Au25L18− (L = S(CH2)2Ph) on the β2-microglobulin natural fibrillation propensity. To reveal the molecular basis of the protein–nanoparticle association process, we performed various simulations at multiple levels (Classical Molecular Dynamics and Brownian Dynamics) that cover multiple length- and timescales. The results provide a model of the ensemble of structures constituting the protein–gold nanoparticle complexes, and insights into the driving forces for the binding of β2-microglobulin to hydrophobic small size gold nanoparticles. We have found that the small nanoparticles can bind the protein to form persistent complexes. This binding of nanoparticles is able to block the active sites of domains from binding to another protein, thus leading to potential inhibition of the fibrillation activity. A comparison with the binding patches identified for the interaction of the protein with a known inhibitor of fibrillation, supports our conclusion.

 Please wait while we load your content...
Please wait while we load your content...