The influence of precursors on phase evolution of nano iron oxides/oxyhydroxides: optical and magnetic properties†
Abstract
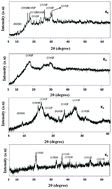
The present investigation describes the evolution of nano iron oxide/oxyhydroxide phases synthesized under identical conditions of precipitation using different starting reagents. Depending on the Fe(III) source and the neutralizing agent, single-phased nanogoethite, single-phased ferrihydrite and a combination of goethite, hematite and ferrihydrite phases were evolved in the synthesized samples. The prepared precursors were annealed at 400, 600 and 800 °C to study the changes in the morphology, optical and magnetic properties of single phase α-Fe2O3. Detailed characterization studies of the precursors and hematite obtained by annealing the samples were carried out using X-ray diffraction (XRD), Transmission electron microscopy (TEM), Raman and Mössbauer spectroscopy. Raman spectra of α-Fe2O3 obtained upon annealing the precursors showed changes in peaks' positions and intensities. These changes have been attributed to surface defects, the phonon confinement effect, the particle shape, and surface morphology. TEM images of the as-synthesized and annealed samples clearly showed differences in the shape, size and crystallinity. The direct and indirect band gap energies of annealed samples estimated from UV-vis absorption spectra showed these values to be smaller as compared to the bulk value for α-Fe2O3. The band gaps were not much affected by the nature of the precursor for the formation of α-Fe2O3 but did depend on annealing temperature. Very interesting trends were observed for the magnetic behaviour of α-Fe2O3. The coercivity values varied in the range of 28.7 to 2966 Oe. Probable mechanisms of formation of different phases have been outlined.

 Please wait while we load your content...
Please wait while we load your content...