The effect of temperature on interhalogen interactions in a series of isostructural organic systems†
Abstract
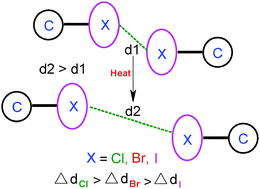
The thermal expansion behaviors of two sets, the guest-free forms and the ethyl acetate solvates, of interhalogen interaction mediated isostructural networks of 2,4,6-triethyl-1,3,5-tris(4-halophenoxy)methylbenzene (halo = Cl, Br and I) have been studied. In both of the series, the order of increase in interhalogen distances with increasing temperature is I⋯I < Br⋯Br < Cl⋯Cl which is actually the reverse order of interhalogen interaction strength.

 Please wait while we load your content...
Please wait while we load your content...