Reversible iodine absorption of nonporous coordination polymer Cu(TCNQ)†
Abstract
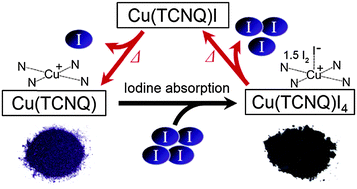
Polycrystalline powders of Cu(TCNQ) (TCNQ = 7,7′,8,8′-tetracyanoquinodimethane) absorb iodine to form Cu(TCNQ)I4 upon solid grinding with iodine or immersion in a hexane solution of iodine. Of the two polymorphs of Cu(TCNQ), phase II Cu(TCNQ) exhibits a much slower iodine-absorption rate than that of phase I Cu(TCNQ) in the liquid-phase reaction, whereas the solid grinding reaction results in efficient absorption for both phases. The valence state of the iodine-containing salt is [Cu+I−(TCNQ0)](I2)1.5, where the copper ion is coordinated with an iodide anion and neutral TCNQ. The salt is a semiconductor (σRT = 3 × 10−3 S cm−1, compaction pellet) with an electrical conductivity one order lower than that of Cu(TCNQ). The salt releases iodine by annealing to regenerate the original phases of Cu(TCNQ) via an intermediate Cu(TCNQ)I state. A solid-state reaction of TCNQ, CuI, and iodine also produces the iodine-containing salt. The iodine absorption–desorption mechanism of Cu(TCNQ) differs from that of alkali-TCNQ salts that we reported previously.

 Please wait while we load your content...
Please wait while we load your content...