Enhancement of hydroxyl radical generation of a solid state photo-Fenton reagent based on magnetite/carboxylate-rich carbon composites by embedding carbon nanotubes as electron transfer channels†
Abstract
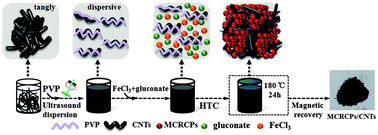
A novel visible-light-driven solid state photo-Fenton reagent based on magnetite/carboxylate-rich carbon particles/carbon nanotubes (MCRCPs–CNTs) has been successfully synthesized by a simple one-pot hydrothermal carbonization (HTC) method using polyvinylpyrrolidone (PVP) as the CNT dispersant. By using PVP as the CNT dispersant, the CNT bundles are well dispersed and interwoven among the MCRCPs, such a structural feature is beneficial for efficient electron transfer. MCRCPs and CNTs play synergistic effects in the photocatalytic process. MCRCPs containing ferricarboxylate complexes act as the solid state photo-Fenton reagent, which realizes the solidification of iron ions and carboxylic acids at the same time. Without using H2O2, MCRCPs containing ferricarboxylate complexes can yield ˙OH under visible light irradiation. By using CNTs as fast electron transfer channels, the photoexcited electrons from the MCRCPs can be quickly trapped by O2 to form very reactive superoxide radical ions (O2˙−) and ultimately convert to hydroxyl radicals (˙OH). The enhanced generation of ˙OH gave the MCRCPs–CNTs advantages over magnetite/carboxylate-rich carbon spheres (MCRCSs) in the photodegradation of methylene blue (MB). In addition, the magnetic saturation (Ms) value is about 24 emu g−1. After the reaction ended, the MCRCPs–CNTs could be rapidly separated under an applied magnetic field.

 Please wait while we load your content...
Please wait while we load your content...