Deletion of the zupT gene for a zinc importer influences zinc pools in Cupriavidus metallidurans CH34
Abstract
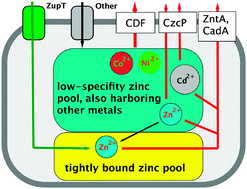
Cupriavidus metallidurans strain CH34 accomplishes a high level of transition metal resistance by a combination of rather unspecific transition metal import and controlled efflux of surplus metals. Using the plasmid-free mutant strain AE104 that possesses only a limited number of metal efflux systems, cellular metal pools were identified as counterparts of these transport reactions. At low zinc concentrations strain AE104 took up Zn(II) until the zinc content reached an optimum level of 70 000 Zn(II) per cell in the exponential phase of growth, whereas a ΔzupT mutant lacking the zinc importer ZupT contained only 20 000 Zn(II)/cell, possibly the minimum zinc content. Mutant and parent cells accumulated up to 125 000 Zn(II) per cell at high (100 μM) external zinc concentrations (optimum zinc content). When the mutant strain Δe4, which has all the known genes for zinc efflux systems deleted, was cultivated in the presence of zinc concentrations close to its upper tolerance level (10 μM), these cells contained 250 000 Zn(II) per cell, probably the maximum zinc content. Instead of zinc, 120 000 cobalt or cadmium ions could also fill-up parts of this zinc pool, showing that it is in fact an undefined pool of divalent transition metal cations bound with low substrate specificity. Even when the cells contained sufficient numbers of total zinc, the zinc importer ZupT was required for important cellular processes, indicating the presence of a pool of tightly bound zinc ions, which depends on ZupT for efficient replenishment. The absence of ZupT led to the formation of inclusion bodies, perturbed oxidative stress resistance and decreased efficiency in the synthesis of the zinc-dependent subunit RpoC of the RNA polymerase, leading to RpoC accumulation. Moreover, when a czc allele for a zinc-exporting transenvelope efflux system CzcCBA was constitutively expressed in a ΔzupT mutant, this led to the disappearance of the CzcA protein and the central subunit of the protein complex, and to the loss of metal resistance. This phenomenon occurred only if the ΔzupT cells had been cultivated for several transfers in solid culture medium, which generated severe zinc starvation. Thus, two zinc pools appear to exist in C. metallidurans: one pool of at least 20 000 tightly bound Zn(II) ions, in addition to a second pool of up to 200 000 cations bound loosely and with low substrate specificity.

 Please wait while we load your content...
Please wait while we load your content...