Beyond substrate analogues: new inhibitor chemotypes for glycosyltransferases
Abstract
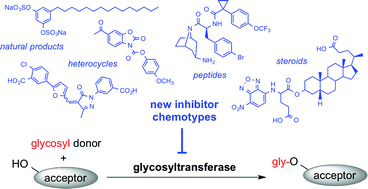
Glycosyltransferases (GTs) are a large family of carbohydrate-active enzymes, which act as nature's glycosylation agents. GTs catalyse the transfer of a mono- or oligosaccharide from a glycosyl donor to an individual acceptor, and play a central role in the biosynthesis of complex carbohydrates, glycans and glycoconjugates. Several GTs have emerged as potential drug targets in a range of therapeutic areas, including infection, inflammation and cancer. Small molecular GT inhibitors are therefore sought after not only as chemical tools for glycobiology, but also as potential lead compounds for drug discovery. Most existing GT inhibitors are donor or acceptor analogues with limited potential for further development due to intrinsic drawbacks, such as a lack of cell penetration and limited chemical stability. In this article, we review recent progress in the identification of alternative inhibitor chemotypes that are not structurally derived from GT donors or acceptors. This growing class of non-substrate-like GT inhibitors now includes several examples with drug-like properties, which provide exciting new starting points for medicinal chemistry and drug discovery. The increasing availability of such alternative GT inhibitor chemotypes represents a significant advance, which will help realise the considerable potential of this important enzyme family as therapeutic targets.

 Please wait while we load your content...
Please wait while we load your content...