Human airway musculature on a chip: an in vitro model of allergic asthmatic bronchoconstriction and bronchodilation†
Abstract
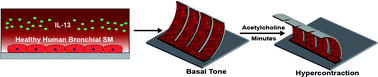
Many potential new asthma therapies that show promise in the pre-clinical stage of drug development do not demonstrate efficacy during clinical trials. One factor contributing to this problem is the lack of human-relevant models of the airway that recapitulate the tissue-level structural and functional phenotypes of asthma. Hence, we sought to build a model of a human airway musculature on a chip that simulates healthy and asthmatic bronchoconstriction and bronchodilation in vitro by engineering anisotropic, laminar bronchial smooth muscle tissue on elastomeric thin films. In response to a cholinergic agonist, the muscle layer contracts and induces thin film bending, which serves as an in vitro analogue for bronchoconstriction. To mimic asthmatic inflammation, we exposed the engineered tissues to interleukin-13, which resulted in hypercontractility and altered relaxation in response to cholinergic challenge, similar to responses observed clinically in asthmatic patients as well as in studies with animal tissue. Moreover, we reversed asthmatic hypercontraction using a muscarinic antagonist and a β-agonist which are used clinically to relax constricted airways. Importantly, we demonstrated that targeting RhoA-mediated contraction using HA1077 decreased basal tone, prevented hypercontraction, and improved relaxation of the engineered tissues exposed to IL-13. These data suggest that we can recapitulate the structural and functional hallmarks of human asthmatic musculature on a chip, including responses to drug treatments for evaluation of safety and efficacy of new drugs. Further, our airway musculature on a chip provides an important tool for enabling mechanism-based search for new therapeutic targets through the ability to evaluate engineered muscle at the levels of protein expression, tissue structure, and tissue function.

 Please wait while we load your content...
Please wait while we load your content...