A microfluidic platform for profiling biomechanical properties of bacteria
Abstract
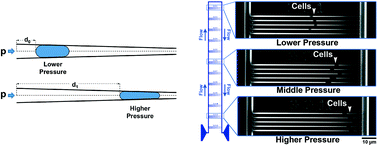
The ability to resist mechanical forces is necessary for the survival and division of bacteria and has traditionally been probed using specialized, low-throughput techniques such as atomic force microscopy and optical tweezers. Here we demonstrate a microfluidic technique to profile the stiffness of individual bacteria and populations of bacteria. The approach is similar to micropipette aspiration used to characterize the biomechanical performance of eukaryotic cells. However, the small size and greater stiffness of bacteria relative to eukaryotic cells prevents the use of micropipettes. Here we present devices with sub-micron features capable of applying loads to bacteria in a controlled fashion. Inside the device, individual bacteria are flowed and trapped in tapered channels. Less stiff bacteria undergo greater deformation and therefore travel further into the tapered channel. Hence, the distance traversed by bacteria into a tapered channel is inversely related to cell stiffness. We demonstrate the ability of the device to characterize hundreds of bacteria at a time, measuring stiffness at 12 different applied loads at a time. The device is shown to differentiate between two bacterial species, E. coli (less stiff) and B. subtilis (more stiff), and detect differences between E. coli submitted to antibiotic treatment from untreated cells of the same species/strain. The microfluidic device is advantageous in that it requires only minimal sample preparation, no permanent cell immobilization, no staining/labeling and maintains cell viability. Our device adds detection of biomechanical phenotypes of bacteria to the list of other bacterial phenotypes currently detectable using microchip-based methods and suggests the feasibility of separating/selecting bacteria based on differences in cell stiffness.

 Please wait while we load your content...
Please wait while we load your content...