Elemental recoveries for metal oxide nanoparticles analysed by direct injection ICP-MS: influence of particle size, agglomeration state and sample matrix
Abstract
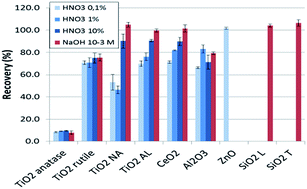
The direct analysis of metal-oxide nanoparticles (NPs) in suspension by inductively coupled plasma-mass spectrometry (ICP-MS) has been investigated. The roles of the chemical nature, size, crystalline form and agglomeration state of the particles in the recovery rate – compared with dissolved samples – have been investigated. Various sample matrices have been tested with the aim to optimize the decomposition process of the particles in the plasma, which governs the quality of the ICP-MS analytical results. It was found that, for SiO2 and ZnO, full recovery was readily obtained in 10−3 mol L−1 NaOH or 0.1% HNO3, respectively. In the case of more stable oxides like TiO2 or CeO2, a positive correlation of the recovery with the concentration of HNO3 in the matrix could be observed, although only NaOH could lead to identical sensitivities for NPs and ionic solutes. Al2O3 could not be satisfactorily analysed (80% recovery at the most). Size and agglomeration state characterization of the selected particles was performed by scanning electron microscopy and dynamic light scattering. The agglomeration state was found to be of minor influence but the size of the primary particle as well as its crystalline phase plays an important role in the analytical recovery.

 Please wait while we load your content...
Please wait while we load your content...