Determination of stability constants of strong metal–ligand complexes using anion or cation exchange chromatography and atomic spectrometry detection†
Abstract
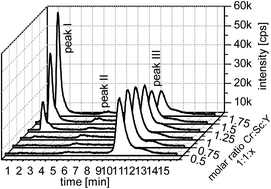
A method for the determination of stability constants of polyaminocarboxylic acid ligands with trivalent metal ions based on the addition of a second metal ion as a competitor has been developed. However, the absolute values of these stability constants are too high for an accurate direct determination. A competition approach delivers the ratio of the stability constants of the two competing metal ions. The different species were separated via anion and cation ion exchange chromatography and their metal content was quantitatively detected online utilizing inductively coupled plasma atomic emission spectrometry. The model ligand used was N,N,N′,N′-ethylenediaminetetraacetic acid as a most prominent polyaminocarboxylic acid ligand. The trivalent ions of chromium, iron, gallium, indium, and scandium were successfully investigated using this approach.
 Please wait while we load your content...
Please wait while we load your content...