Insights into biphasic oxidations with hydrogen peroxide; towards scaling up
Abstract
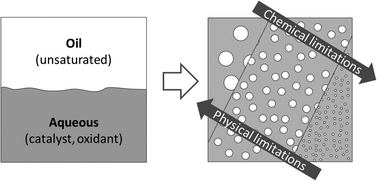
Bi-phasic oxidations using hydrogen peroxide oxidant and a tungsten-based catalyst (e.g. Na2WO4) can proceed quickly and effectively without the need for organic solvents or added phase transfer agents in emulsified systems. Providing sufficient contact area between phases easily overcomes the physical limitations associated with the phase transfer of species, facilitating its scale-up, recyclability of the catalyst and easing product separation. Using the epoxidation of sunflower seed oil as a model reaction, we have also shown how the chemical and safety limitations associated with the parallel decomposition of hydrogen peroxide by Na2WO4 can be suppressed by the use of readily available water soluble organic carboxylic acids and careful consideration of the catalyst to acid ratio. The combination of chemical and engineering solutions with a near 100% hydrogen peroxide use, makes, for the first time, this green oxidation pathway attractive for large-scale industrial applications.

 Please wait while we load your content...
Please wait while we load your content...