Catalyst-free direct arylsulfonylation of N-arylacrylamides with sulfinic acids: a convenient and efficient route to sulfonated oxindoles†
Abstract
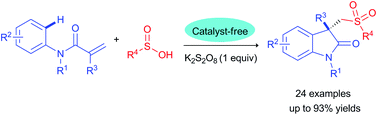
A simple, efficient and catalyst-free procedure has been developed for the construction of sulfonated oxindoles via the direct arylsulfonylation of N-arylacrylamides with sulfinic acids. The present protocol, which simply utilizes cheap oxidants, readily-available starting materials, and catalyst-free conditions, provides an alternative and highly attractive approach to a series of sulfonated oxindoles with high atom efficiency and excellent functional group tolerance.

 Please wait while we load your content...
Please wait while we load your content...