Engineering cyanobacteria for the production of a cyclic hydrocarbon fuel from CO2 and H2O
Abstract
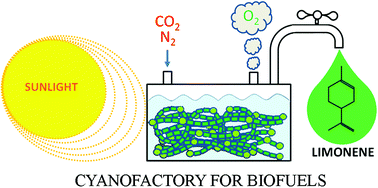
Cyclic hydrocarbons are a critical component of petroleum fuels. However, biofuels produced by current biochemical and thermochemical processes contain small amounts of cyclic hydrocarbons, and can only provide the requisite performance characteristics with the addition of petroleum fuels to them. Limonene (C10H16) is a cyclic monoterpene that possesses attractive characteristics as a biodiesel and a jet fuel. Current strategies for harvesting limonene from plant biomass require arable land, high energy input, inefficient multiple-step processes, and the release of CO2 as a greenhouse gas. This research focuses on a direct photons-to-product approach for biofuel production by metabolically engineering a cyanobacterium as the cellular machinery to over-produce and secrete valuable compounds using CO2, mineralized H2O, and light. As a proof of concept, we have engineered the filamentous, nitrogen-fixing cyanobacterium Anabaena sp. PCC 7120 to synthesize and secrete limonene by introducing a plant limonene synthase gene (lims) from Sitka spruce. Our data revealed that limonene produced by the engineered cyanobacterium was secreted across the cell membrane and volatilized into the headspace, allowing for easy separation of the target compound from the culture biomass. Furthermore, a synthetic DXP operon (dxs-ipphp-gpps) encoding three rate-limiting enzymes from the MEP pathway was co-expressed with lims to re-route carbon flux from the Calvin cycle into limonene synthesis. Under higher light (150 μE m−2 s−1), we observed a 6.8-fold increase in limonene yield and an 8.8-fold increase in the maximum limonene production rate when expressing the DXP operon in conjunction with lims, compared to lims alone, and achieved a maximum production rate of 3.6 ± 0.5 μg limonene L−1 O.D.−1 h−1. This limonene-producing Anabaena has about three times higher photosystem II activity than its wild-type. These results demonstrated that increasing the light intensity and metabolic flux improves limonene productivity in the engineered cyanobacterium. We envision that the method of using N2-fixing cyanobacteria as a cellular factory and CO2 and N2 as sustainable feedstock can be applied for the production of a wide range of commodity chemicals and drop-in-fuels.

 Please wait while we load your content...
Please wait while we load your content...