Effect of natural organic matter on the disagglomeration of manufactured TiO2 nanoparticles†
Abstract
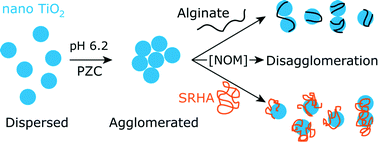
One major concern in the fate of nanomaterials in aquatic systems is the lack of data on nanomaterial transformations under relevant environmental conditions. The disagglomeration of aggregates composed of manufactured anatase titanium dioxide nanoparticles is investigated here in the presence of alginate and Suwannee River humic acids at varying concentrations using dynamic light scattering and electrophoretic measurements. Stability of TiO2 nanoparticle agglomerates at typical environmental concentrations of natural organic matter is discussed at a pH value corresponding to the point of zero charge of TiO2 nanoparticles. In this scenario, the surface charge of TiO2 is neutralized, allowing the nanoparticles to form large agglomerates. Alginate and Suwannee River humic acids exhibit a negative structural charge under this pH condition and adsorption of both natural polyelectrolytes on the surface of nanoparticle agglomerates leads to disagglomeration and significant redispersion of TiO2 nanoparticles into fragments. Results indicate that both electrostatic forces and steric interactions play key roles during the disagglomeration process and that the physicochemical properties of natural organic matter are found to influence the kinetics and importance of fragmentation in the disagglomeration process. Most importantly, our data indicate that the presence of natural organic matter at typical environmental concentrations induces significant disagglomeration of large submicron nanoparticle agglomerates. Such a result constitutes an important outcome with regards to the risk associated with manufactured nanoparticles by including the possible transformations of the micron size range structures they can form.

 Please wait while we load your content...
Please wait while we load your content...