Natural water chemistry (dissolved organic carbon, pH, and hardness) modulates colloidal stability, dissolution, and antimicrobial activity of citrate functionalized silver nanoparticles†
Abstract
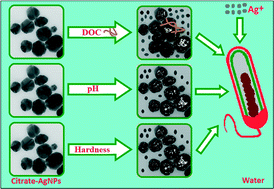
Knowledge about whether/how natural water chemistry influences the fate, dissolution, and toxicity of silver nanoparticles (AgNPs) should contribute to ecological risk assessment and informed decision making. The effects of three critical water chemistry parameters – dissolved organic carbon (DOC), pH, and hardness – were investigated on the colloidal stability, dissolution dynamics, and antimicrobial activity of citrate-functionalized AgNPs (citrate–AgNPs) against Escherichia coli. Toxicities of citrate–AgNPs and AgNO3 were also determined in the river water samples collected across three seasons (for seven months). Detectable changes in hydrodynamic diameter, surface charge, and plasmonic resonance revealed the modulating effects of the water chemistry parameters on the colloidal stability of citrate–AgNPs. Although, overall Ag release from citrate–AgNPs was low (0.33–3.62%), it increased with increasing DOC concentrations (0–20 mg L−1) but decreased with increasing pH (5–7.5) or hardness (150–280 mg L−1). Citrate–AgNP toxicity was 3–44 fold lower than of AgNO3 (Ag mass basis). Notably, higher DOC or pH conferred protection to E. coli against citrate–AgNPs or AgNO3; increasing solution hardness tended to enhance toxicity, however. Citrate–AgNPs or AgNO3 toxicity in the river water matrix revealed no seasonality. Generalized linear models developed, by parameterizing particle properties, could fairly predict empirically-derived nanotoxicity. Our results show that particle size, surface properties, ion release kinetics, and toxicity of citrate–AgNPs can be modified upon release into aquatic environments, suggesting potential implications to ecosystem health and functions.
- This article is part of the themed collection: Environmental Science: Nano 2014 Most Accessed Articles

 Please wait while we load your content...
Please wait while we load your content...