Shale gas-to-syngas chemical looping process for stable shale gas conversion to high purity syngas with a H2 : CO ratio of 2 : 1
Abstract
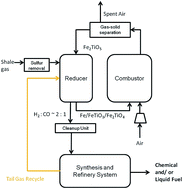
The shale gas-to-syngas (STS) chemical looping process was conceived by Fan and associates in 2013 for the production of high-purity syngas from shale gas. The STS process producing syngas does not require the use of molecular oxygen from air separation and steam. This paper describes the rationale for the process concept with experimental data that substantiates the process validity. Specifically, the STS process consists of a co-current gas (shale gas)–solid (metal oxides) moving bed contact mode reducer operation with metal oxides for shale gas conversion to syngas. The reduced metal oxides from the reducer operation are regenerated via an oxidation operation with air. Various active metal oxides or metal oxide composites can be utilized. However, it is through the combination of desired metal oxides and co-current moving bed reducer that high syngas purity and a desirable H2 : CO molar ratio of ∼2 : 1 can be achieved. In this study, active iron–titanium composite metal oxide (ITCMO) materials are used as the oxygen carrier for the demonstration of the STS process. The desirable thermodynamic property of ITCMO is a key factor for the generation of high quality syngas. The co-current moving bed provides a desirable gas–solid contacting pattern that minimizes carbon deposition and maximizes the syngas yield. The syngas produced by the STS process can achieve a H2 : CO molar ratio of ∼2 : 1 with little CO2, CH4 and steam, which is required for downstream processes to produce liquid fuels and chemicals. The experimental results for reaction kinetics including oxygen carrier recyclability and pressure effects are obtained by thermogravimetric analysis (TGA), and syngas generation using a fixed bed, a bench-scale moving bed, and a sub-pilot scale moving bed reactor demonstrations are achieved in this study. The bench and sub-pilot demonstrations confirm that the syngas produced by the STS process is close to thermodynamic equilibrium with the reduced ITCMO. Furthermore, simulation studies are conducted to compare the efficiency of the STS process with a conventional autothermal natural gas reforming process.

 Please wait while we load your content...
Please wait while we load your content...