Enhanced photoelectrochemical water-splitting performance of semiconductors by surface passivation layers
Abstract
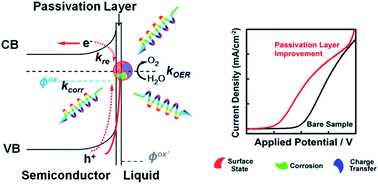
An important approach for solving the world's sustainable energy challenges is the conversion of solar energy to chemical fuels. Semiconductors can be used to convert/store solar energy to chemical bonds in an energy-dense fuel. Photoelectrochemical (PEC) water-splitting cells, with semiconductor electrodes, use sunlight and water to generate hydrogen. Herein, recent studies on improving the efficiency of semiconductor-based solar water-splitting devices by the introduction of surface passivation layers are reviewed. We show that passivation layers have been used as an effective strategy to improve the charge-separation and transfer processes across semiconductor–liquid interfaces, and thereby increase overall solar energy conversion efficiencies. We also summarize the demonstrated passivation effects brought by these thin layers, which include reducing charge recombination at surface states, increasing the reaction kinetics, and protecting the semiconductor from chemical corrosion. These benefits of passivation layers play a crucial role in achieving highly efficient water-splitting devices in the near future.

 Please wait while we load your content...
Please wait while we load your content...