Revisiting film theory to consider approaches for enhanced solvent-process design for carbon capture
Abstract
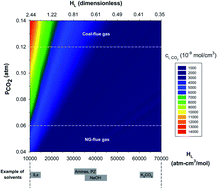
Application of carbon capture at the gigaton-scale necessary for significant reduction in atmospheric CO2 requires a portfolio of technologies for applications that may span point-source capture to more dilute systems such as CO2 removal from the atmosphere. We argue that for absorption separation processes there is a strong coupling between the solvent and process properties, which are uniquely dependent upon the starting concentration of CO2. We revisit Whitman's film theory and consider mass-transfer correlations to determine the most critical solvent and process parameters that influence the flux of CO2 from the gas to the liquid phase, within which it is ultimately captured. Finally, results of this work indicate, for instance, that increasing the kinetics of a reacting solvent with CO2 has a greater impact on direct air capture (DAC) systems, than natural gas- or coal-fired power plant emissions. In addition, the solvent kinetics is a more influential parameter than the Henry's law solubility coefficient for DAC systems, while the reverse may be found for more concentrated CO2 gas mixtures.

 Please wait while we load your content...
Please wait while we load your content...