Complexity in seemingly simple sodium magnesiate systems†
Abstract
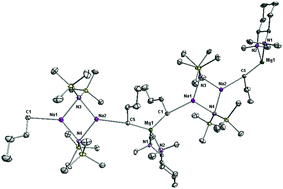
A systematic study both in the solid- and solution-state, was carried out for a series of sodium magnesiates containing the utility amide ligand 1,1,1,3,3,3-hexamethyldisilazide (HMDS). The first complex considered is the donor-free bisamido monoalkyl polymeric complex [Na(μ-HMDS)2Mg(nBu)]∞1. The reactivity of 1 with common tertiary bidentate donors including N,N,N′,N′-tetramethylethylenediamine (TMEDA) or its chiral relative (1R,2R)-tetramethylcyclohexyldiamine [(R,R)-TMCDA] is detailed. Surprisingly, the products of these reactions are not simple diamine adducts but are solvent separated sodium magnesiate systems [(TMEDA)2·Na]+[Mg(HMDS)3]−2 and [{(R,R)-TMCDA}2·Na]+[Mg(HMDS)3]−3. By concentrating on the likely equilibria which may give rise to formation of 2, a potential intermediate complexed ion pair [{(TMEDA)2·Na}(μ-nBu)Mg(HMDS)2] 4 was isolated. Additionally, the novel “inverse magnesiates” [{Na(μ-HMDS)}2Mg(μ-nBu)2·(TMEDA)]∞5 and [{Na(μ-HMDS)}2Mg(μ-nBu)2·{(R,R)-TMCDA}]∞6, were obtained by reacting solutions of composition “NaMg(HMDS)(nBu)2” (a likely by-product in the formation of 2 from 1), with TMEDA or (R,R)-TMCDA. The structure and nature of these bimetallic complexes have been determined using a combination of X-ray crystallographic studies and multinuclear NMR spectroscopy.
- This article is part of the themed collection: New Expeditions in Polar Organometallic Chemistry

 Please wait while we load your content...
Please wait while we load your content...