A comparative synthetic, magnetic and theoretical study of functional M4Cl4 cubane-type Co(ii) and Ni(ii) complexes†
Abstract
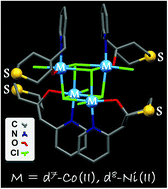
We describe the synthesis, structures, and magnetochemistry of new M4Cl4 cubane-type cobalt(II) and nickel(II) complexes with the formula [M(μ3-Cl)Cl(HL·S)]4 (1: M = Co; 2: M = Ni), where HL·S represents a pyridyl-alcohol-type ligand with a thioether functional group, introduced to allow subsequent binding to Au surfaces. Dc and ac magnetic susceptibility data of 1 and 2 were modeled with a full spin Hamiltonian implemented in the computational framework CONDON 2.0. Although both coordination clusters 1 and 2 are isostructural, with each of their transition metal ions in a pseudo-octahedral coordination environment of four Cl atoms and N,O-donor atoms of one chelating HL·S ligand, the substantially different ligand field effects of Co(II) and Ni(II) results in stark differences in their magnetism. In contrast to compound 1 which exhibits a dominant antiferromagnetic intramolecular coupling (J = −0.14 cm−1), 2 is characterised by a ferromagnetic coupling (J = +10.6 cm−1) and is considered to be a single-molecule magnet (SMM), a feature of special interest for future surface deposition studies. An analysis based on density functional theory (DFT) was performed to explore possible magnetostructural correlations in these compounds. Using a two-J model Hamiltonian, it revealed that compound 1 has four positive and two (small) negative JCo⋯Co isotropic interactions leading to a SHS = 6 ground state. Taking into account the magnetic anisotropy, one would recover a MS = 0 ground state since D > 0 from computations. In 2, all the J constants are positive and, in this framework, the zero-field splitting energy characterising the axial anisotropy was estimated to be negative (D = −0.44 cm−1). The computational results are consistent with compound 2 being an SMM.

 Please wait while we load your content...
Please wait while we load your content...