Peptide-based FeS4 complexes: the zinc ribbon fold is unsurpassed to stabilize both the FeII and FeIII states†
Abstract
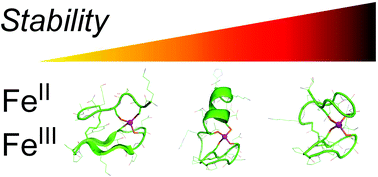
Whereas Zn(Cys)4 zinc fingers exist with different protein folds, only the zinc ribbon fold is found in rubredoxin Fe(Cys)4 sites. To assess the significance of this observation, we have investigated the binding and stability of Fe2+ and Fe3+ ions by a set of four peptides designed to model Zn(Cys)4 zinc fingers with various folds, i.e. zinc ribbon, treble clef and a loosened zinc ribbon fold. All peptides were shown by means of UV-Vis and CD spectroscopies to form stable 1 : 1 FeII/peptide complexes with binding constants higher than 107 M−1 at pH 7. Their oxidation into FeIII complexes and the stability of the latter were compared. The UV-Vis absorption and CD spectroscopic properties of the FeII and FeIII complexes were analysed with respect to the structures of the zinc analogues in order to get insight into the local arrangement of the Fe(Cys)4 core around the metal ion. The chemical stability of these complexes was rationalized according to the shielding from the solvent provided by the various peptide folds to the FeS4 core. In addition, we showed that whereas UV-visible spectra inform only on the FeS4, the information derived from the corresponding CD spectra extend to the Cβ orientation and the peptide fold. The results presented here demonstrate that while the zinc ribbon fold is not strictly required to obtain a Fe(Cys)4 site, it affords a drastically superior protection of the site toward external redox agents. This finding brings new clues to engineer stable and redox-active Fe(Cys)4 sites in de novo proteins.

 Please wait while we load your content...
Please wait while we load your content...