Abstract
Mono- and dianionic carboranylamidinates are readily available in one-pot reactions directly from o-carborane (1). In situ-monolithiation of 1 followed by treatment with N,N′-diisopropylcarbodiimide, iPrN![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NiPr, or N,N′-dicyclohexylcarbodiimide, CyN
NiPr, or N,N′-dicyclohexylcarbodiimide, CyN![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NCy, provided the lithium carboranylamidinates (o-C2B10H10C(NHiPr)(
NCy, provided the lithium carboranylamidinates (o-C2B10H10C(NHiPr)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NiPr)-κ2C,N)Li(DME) (2a) and (o-C2B10H10C(NHiCy)(
NiPr)-κ2C,N)Li(DME) (2a) and (o-C2B10H10C(NHiCy)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NiCy)-κ2C,N)Li(THF)2 (2b). Controlled hydrolysis of 2a,b afforded the free carboranylamidines o-C2B10H11C(NHiR)(
NiCy)-κ2C,N)Li(THF)2 (2b). Controlled hydrolysis of 2a,b afforded the free carboranylamidines o-C2B10H11C(NHiR)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NiR) (3a: R = iPr, 3b: R = Cy). The first dimetalated carboranylamidinates, o-C2B10H10C(NiPr)(
NiR) (3a: R = iPr, 3b: R = Cy). The first dimetalated carboranylamidinates, o-C2B10H10C(NiPr)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NiPr)Li2(DME)2 (4a) (DME = 1,2-dimethoxyethane) and o-C2B10H10C(NiPr)(
NiPr)Li2(DME)2 (4a) (DME = 1,2-dimethoxyethane) and o-C2B10H10C(NiPr)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NiPr)Li2(THF)4 (4b), were prepared in high yield (83% yield) directly from 1 using a simple one-pot synthetic protocol. Treatment of 4b with 2 equiv. of Me3SiCl afforded the disilylated derivative o-C2B10H10-κ2C,N-[C(NiPrSiMe3)(
NiPr)Li2(THF)4 (4b), were prepared in high yield (83% yield) directly from 1 using a simple one-pot synthetic protocol. Treatment of 4b with 2 equiv. of Me3SiCl afforded the disilylated derivative o-C2B10H10-κ2C,N-[C(NiPrSiMe3)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NiPr)]SiMe3 (5). Dianionic 4b also served as an excellent precursor for novel inorganic heterocycles incorporating the closo-1,2-C2B10H10 cage, including the unsymmetrical distannene [o-C2B10H10C(NiPr)(
NiPr)]SiMe3 (5). Dianionic 4b also served as an excellent precursor for novel inorganic heterocycles incorporating the closo-1,2-C2B10H10 cage, including the unsymmetrical distannene [o-C2B10H10C(NiPr)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NiPr)-κ2C,N]Sn
NiPr)-κ2C,N]Sn![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Sn[(iPrN)2CnBu]2 (6) and the azaphosphole derivative [o-C2B10H10C(NiPr)(
Sn[(iPrN)2CnBu]2 (6) and the azaphosphole derivative [o-C2B10H10C(NiPr)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NiPr)-κ2C,N]PPh (7). Surprisingly, it was found that the synthesis of new inorganic ring systems from dianionic carboranylamidinates can also be achieved by employing only 1 equiv. of n-butyllithium in the generation of the anionic carboranylamidinate intermediates. Using this straightforward one-pot synthetic protocol, the Group 14 metallacycles [o-C2B10H10C(NCy)(
NiPr)-κ2C,N]PPh (7). Surprisingly, it was found that the synthesis of new inorganic ring systems from dianionic carboranylamidinates can also be achieved by employing only 1 equiv. of n-butyllithium in the generation of the anionic carboranylamidinate intermediates. Using this straightforward one-pot synthetic protocol, the Group 14 metallacycles [o-C2B10H10C(NCy)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NCy)-κ2C,N]SiR2 (R = Cl (8), Me (9), Ph (10)) and [o-C2B10H10C(NCy)(
NCy)-κ2C,N]SiR2 (R = Cl (8), Me (9), Ph (10)) and [o-C2B10H10C(NCy)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NCy)-κ2C,N]GeCl2 (11) have become accessible. The same synthetic strategy could be successfully adapted to prepare the corresponding Group 4 metallocene derivatives Cp2Ti[o-C2B10H10C(NCy)(
NCy)-κ2C,N]GeCl2 (11) have become accessible. The same synthetic strategy could be successfully adapted to prepare the corresponding Group 4 metallocene derivatives Cp2Ti[o-C2B10H10C(NCy)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NCy)-κ2C,N] (12) and Cp2Zr[o-C2B10H10C(NCy)(
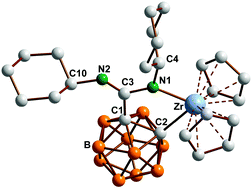
NCy)-κ2C,N] (12) and Cp2Zr[o-C2B10H10C(NCy)(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NCy)-κ2C,N] (13). The molecular structures of 2b, 3b, 4b, 5, 6, 7, 10, 12, and 13 were confirmed by single-crystal X-ray diffraction.
NCy)-κ2C,N] (13). The molecular structures of 2b, 3b, 4b, 5, 6, 7, 10, 12, and 13 were confirmed by single-crystal X-ray diffraction.
- This article is part of the themed collection: Carboranes

 Please wait while we load your content...
Please wait while we load your content...