Structure of a uranyl peroxo complex in aqueous solution from first-principles molecular dynamics simulations†
Abstract
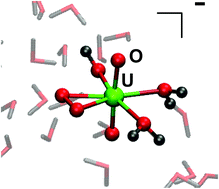
Static ab initio and density-functional computations, as well as Car–Parrinello molecular dynamics simulations in aqueous solution are reported for [UO2(OH)(κ2-O2)(H2O)2]− and [UO2(OH)2(κ1-O2H)(H2O)]−. Whereas the κ1-hydroperoxo isomer is found to be more stable than the κ2-peroxo form in the gas phase, the order of stability is reversed in explicit bulk solution. Based on free energies from thermodynamic integration (BLYP functional), the peroxo form is favoured by ca. 32 kJ mol−1 in water. This stabilisation is discussed in terms of the hydration shells about the individual ligands and dipole moments of the complexes in water, and highlights the importance of explicit solute–solvent interactions and bulk solvation for the speciation of uranyl(VI) compounds.
- This article is part of the themed collection: Synergy between Experiment and Theory

 Please wait while we load your content...
Please wait while we load your content...