Synthesis of new derivatives of copper complexes of Josiphos family ligands for applications in asymmetric catalysis†
Abstract
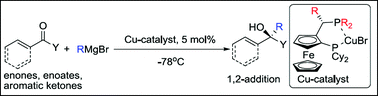
A series of new copper complexes containing chiral ferrocenyl diphosphine ligands of the Josiphos family have been prepared. These complexes have been studied in the catalytic asymmetric 1,2-addition of Grignard reagents to enones and aromatic ketones. Variation of the electronic and steric properties of the ligand resulted in a positive effect in the regio- and enantioselectivity of Grignard reagents to α-H-substituted enones using the ligand in which tert-butyl substituents were introduced in the diarylphosphine moiety. The copper complexes were also successfully applied in the catalytic asymmetric conjugate addition of Grignard reagents to enoates. No increase of enantioselectivity was observed in the catalytic asymmetric addition of linear Grignard reagents, compared to that of the commercially available ligand rev-Josiphos.

 Please wait while we load your content...
Please wait while we load your content...