Effect of TiO2 crystal structure on the catalytic performance of Co3O4/TiO2 catalyst for low-temperature CO oxidation
Abstract
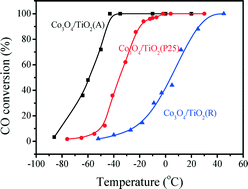
Co3O4 catalysts supported on TiO2 with different crystalline structures (anatase (A), rutile (R) and P25 (Degussa)) were prepared by a deposition–precipitation method, and characterized by nitrogen adsorption/desorption, XRD, HR-TEM, EPR, Raman spectroscopy, XPS and H2-TPR techniques. The results show that Co3O4/TiO2 (A) exhibited the highest activity among the three Co3O4/TiO2 catalysts: CO can be completely oxidized to CO2 at −43 °C. When rutile TiO2 or P25 were used as the support, its catalytic activity was decreased obviously, because the TiO2 crystal structure has an influence on the physicochemical and catalytic properties of the Co3O4 catalysts. The results show that the Co3O4/TiO2 (A) catalyst contains Ti3+ species, which is in an unstable state and can affect the properties of Co3O4 by the interaction between the deposited Co3O4 and anatase TiO2 support. The Co3O4/TiO2 (A) catalyst exhibits highly defective structure and good oxygen adsorption ability. The reducibility of Co3O4 is improved by the anatase TiO2 support, resulting in Co3O4/TiO2 (A) possessing the better redox property than the other Co3O4/TiO2 catalysts, which is an important factor for its high catalytic activity.

 Please wait while we load your content...
Please wait while we load your content...