In situ X-ray Raman spectroscopy study of the hydrogen sorption properties of lithium borohydride nanocomposites†
Abstract
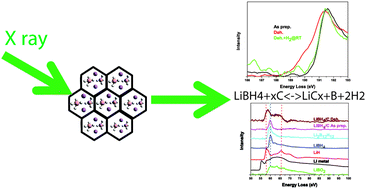
Nanoconfined alkali metal borohydrides are promising materials for reversible hydrogen storage applications, but the characterization of hydrogen sorption in these materials is difficult. Here we show that with in situ X-ray Raman spectroscopy (XRS) we can track the relative amounts of intermediates and final products formed during de- and re-hydrogenation of nanoconfined lithium borohydride (LiBH4) and therefore we can possibly identify the de- and re-hydrogenation pathways. In the XRS of nanoconfined LiBH4 at different points in the de- and re-hydrogenation, we identified phases that lead to the conclusion that de- and re-hydrogenation pathways in nanoconfined LiBH4 are different from bulk LiBH4: intercalated lithium (LiCx), boron and lithium hydride were formed during de-hydrogenation, but as well Li2B12H12 was observed indicating that there is possibly some bulk LiBH4 present in the nanoconfined sample LiBH4–C as prepared. Surprisingly, XRS revealed that the de-hydrogenated products of the LiBH4–C nanocomposites can be partially rehydrogenated to about 90% of Li2B12H12 and 2–5% of LiBH4 at a mild condition of 1 bar H2 and 350 °C. This suggests that re-hydrogenation occurs via the formation of Li2B12H12. Our results show that XRS is an elegant technique that can be used for in and ex situ study of the hydrogen sorption properties of nanoconfined and bulk light-weight metal hydrides in energy storage applications.

 Please wait while we load your content...
Please wait while we load your content...