New understanding of the difference of photocatalytic activity among anatase, rutile and brookite TiO2
Abstract
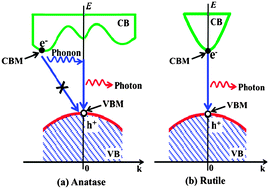
In general, anatase TiO2 exhibits higher photocatalytic activities than rutile TiO2. However, the reasons for the differences in photocatalytic activity between anatase and rutile are still being debated. In this work, the band structure, density of states, and effective mass of photogenerated charge carriers for anatase, rutile and brookite TiO2 are investigated by the first-principle density functional theory calculation. The results indicate that anatase appears to be an indirect band gap semiconductor, while rutile and brookite belong to the direct band gap semiconductor category. Indirect band gap anatase exhibits a longer lifetime of photoexcited electrons and holes than direct band gap rutile and brookite because the direct transitions of photogenerated electrons from the conduction band (CB) to valence band (VB) of anatase TiO2 is impossible. Furthermore, anatase has the lightest average effective mass of photogenerated electrons and holes as compared to rutile and brookite. The lightest effective mass suggests the fastest migration of photogenerated electrons and holes from the interior to surface of anatase TiO2 particle, thus resulting in the lowest recombination rate of photogenerated charge carriers within anatase TiO2. Therefore, it is not surprising that anatase usually shows a higher photocatalytic activity than rutile and brookite. This investigation will provide some new insight into understanding the difference of photocatalytic activity among anatase, rutile and brookite.

 Please wait while we load your content...
Please wait while we load your content...