Theoretical two-atom thick semiconducting carbon sheet
Abstract
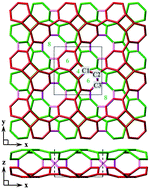
A two-dimensional carbon allotrope, H-net, is proposed using first principle calculations. H-net incorporates C4 distorted squares, C6 hexagons, and C8 octagons. Unlike previously reported planar graphene and other theoretical carbon sheets, H-net is a two-atom thick polymorph with identical C6 + C4 + C6 components cross-facing and covalently buckled to feature a handshake-like model. The feasibility of H-net is evident from its dynamic stability as confirmed by phonon-mode analysis and its lower total energy. H-net is energetically more favorable than synthesized graphdiyne and theoretical graphyne, BPC, S-graphene, polycyclic net, α-squarographite, and lithographite. We explored a possible route for the synthesis of H-net from graphene nanoribbons. Electronic band structure calculations indicated that H-net is a semiconductor with an indirect band gap of 2.11 eV, whereas graphene and many other two-dimensional carbon sheets are metallic. We also explored the electronic structure of one-dimensional nanoribbons derived from H-net. The narrowest H-net nanoribbon showed metallic behavior, whereas the other nanoribbons are semiconductors with band gaps that increase as the nanoribbons widen. H-net and its tailored nanoribbons are expected to possess more electronic properties than graphene because of their exceptional crystal structure and different energy band gaps.

 Please wait while we load your content...
Please wait while we load your content...